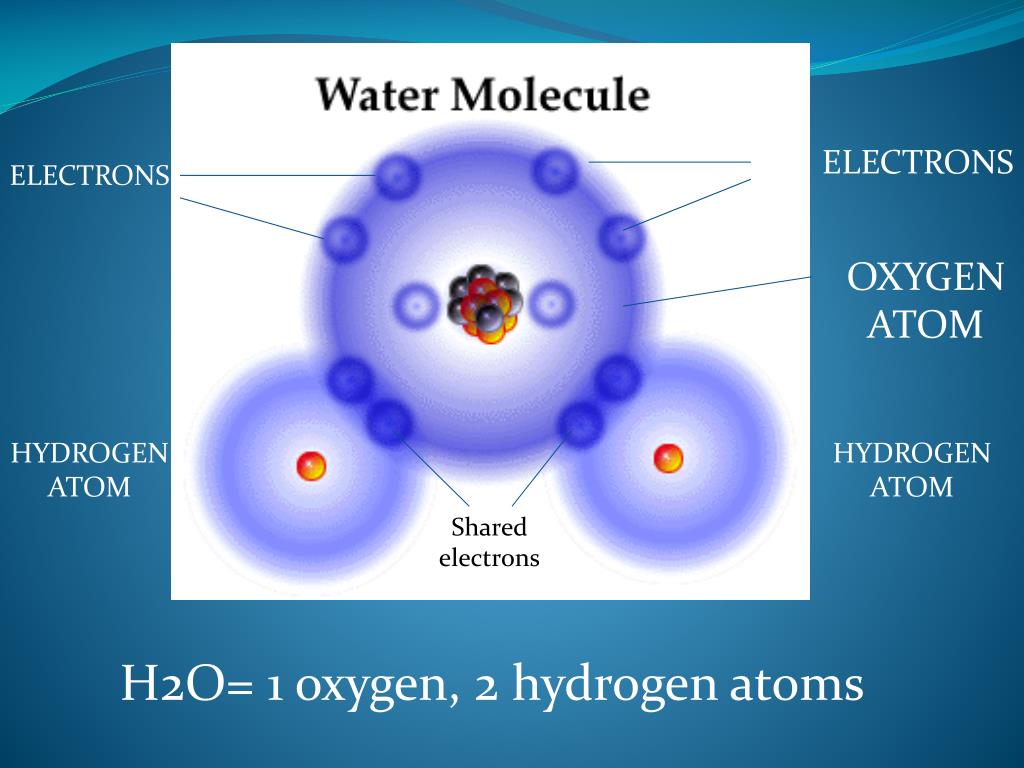  
For example, hydrogen and oxygen atoms combine in a ratio of 2:1 to form. Names for Hydrogen Atoms, Ions, and Groups, and for Reactions Involving Them (Recommendations 1988). Atoms of different elements combine in small, whole-number ratios to form compounds. (2) IUPAC-IUBMB Joint Commission on Biochemical Nomenclature (JCBN) and Nomenclature Committee of IUBMB (NC-IUBMB). Proto- and proti- would be reserved for the $\ce$ nucleus - the proton - that is most commonly interrogated, although deuterium NMR is very much possible. The ratio of atoms we will need to make any number of water molecules is the same: 2 hydrogen atoms to 1 oxygen atom. Under IUPAC's nomenclature proposal, the prefixes hydro- and hydri- would be used to refer to the mixture of hydrogen isotopes that occurs in nature. IUPAC gave some attention to the issue of hydrogen nomenclature and provided guidelines: If a hydrogen atom loses its electron then it is then usually referred to as a proton, although it might be something else depending on the hydrogen isotope involved. Electrons may display a preference for some parts of a molecule, such as the regions between nuclei, so you could say they "hang out" there more. In molecules electrons are usually "shared". Individual H2O molecules are V-shaped, consisting of two hydrogen atoms (depicted in white) attached to the sides of a single oxygen atom (depicted in red). You make some false assumptions, for instance that in water hydrogen atoms give away their electrons and oxygen ends up with 10 electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
